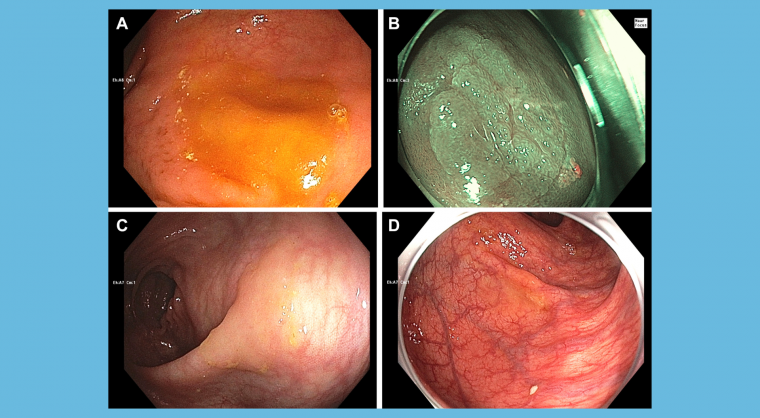
Colonoscopy is the most effective visceral cancer prevention tool in clinical medicine, but its potential is reduced by extreme operator dependence. Recent data indicate that protection against postcolonoscopy colorectal cancer (PCCRC) by colonoscopy continues to improve up to adenoma detection rates (ADRs) of 50%. Thus, most practicing colonoscopists could provide better cancer protection to patients by improving their ADRs.
Improvements in colonoscopist ADR through training and education result in improved CRC protection. Various colonoscopic techniques (eg, double right colon examination, patient position changes during withdrawal, chromoendoscopy) and devices (electronic chromoendoscopy, distal colonoscope attachments and artificial intelligence) also improve ADR.
Although it is widely assumed that ADR increases achieved through ancillary techniques and devices lead to reductions in PCCRC, this remains unproven.
CADe is generally faster than human endoscopists, with shorter time to lesion recognition in more than 80% of lesions.
There are uncertainties about the value of CADe.
- First, CADe increases ADR without addressing mucosal exposure, and AI programs improving mucosal exposure have had more impact on ADR than CADe.
- Further, CADe could increase ADR through detection of primarily small or diminutive adenomas, depending on the lesion set used to train the CADe algorithm. Transparency regarding the data sets used to train the first or upgraded versions of CADe software is often lacking.
- Third, the absolute levels of ADR achieved in randomized trials using AI have been exceeded by some endoscopists performing colonoscopy without AI, though the indications for colonoscopy and the study populations varied between trials.
Read the full article in Gastroenterology from authors Douglas K. Rex, Yuichi Mori, Prateek Sharma, Rachel E. Lahr, Krishna C. Vemulapalli and Cesare Hassan for details on the testing and evaluation of the first CADe program approved by the FDA.