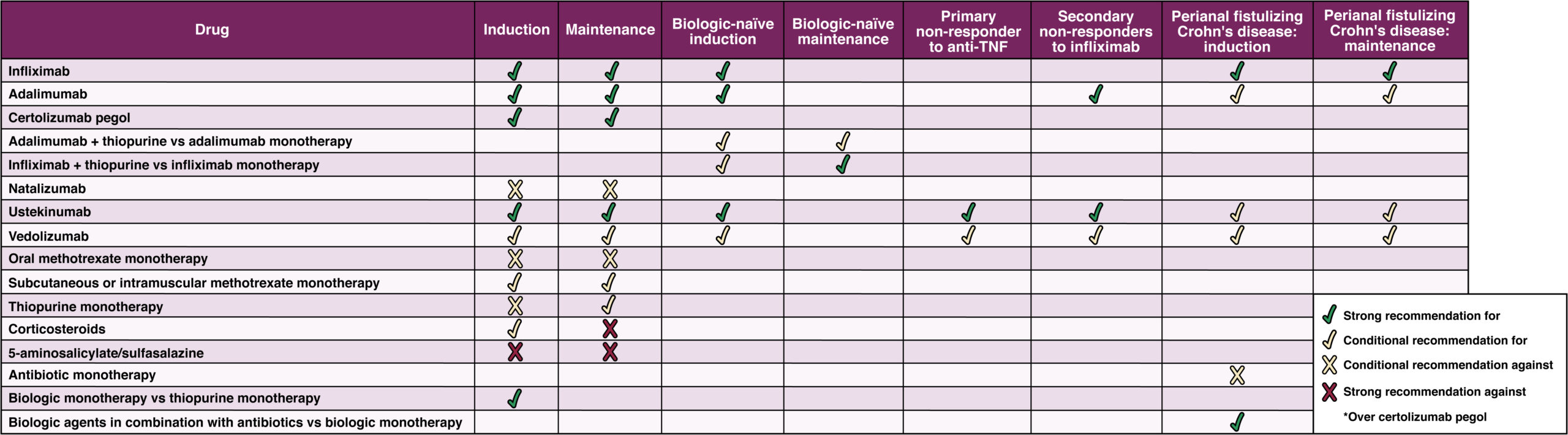
During the past decade, the armamentarium of therapeutic agents for Crohn’s disease has expanded, with new data regularly emerging on safety and efficacy of agents new and old. AGA has just released new clinical practice guidelines evaluating therapies for luminal and fistulizing Crohn’s disease. The guidelines assessed the overall benefit of each drug and when possible compared drugs via a network meta-analysis to assess their efficacy relative to each other. The guideline also recommended against a number of therapies that either lacked evidence or risked patient harm, for example natalizumab.
Main takeaways
Biologics are the most effective drugs for the management of Crohn’s and they should be used early, rather than delaying their use until after failure of mesalamine and/or corticosteroids, in patients with moderate to severe or fistulizing Crohn’s disease.
- Anti-tumor necrosis factor (anti-TNF) agents or ustekinumab are recommended and vedolizumab is suggested as a first-line treatment.
- In patients who have previously not responded to anti-TNF agents, AGA recommends ustekinumab or vedolizumab.
- The biologic natalizumab is no longer recommended due to potential adverse events and the availability of safer treatment options.
Resources
Review the following resources for additional details on how to implement these new guidelines. Have questions? The authors will be available in the AGA Community the week of June 14 to answer any questions you have about applying these guidelines in practice.
- Guidelines
- Technical review
- Spotlight (infographic)
- Clinical decision support tool
- Commentary: Providing the Best Care for Patients with Crohn’s Disease: An Examination of the New AGA Clinical Practice Guideline on the Medical Management of Moderate to Severe Luminal and Fistulizing Crohn’s Disease
The authors will be available in the AGA Community the week of June 14 to answer any questions you have about applying these guidelines in practice. Follow the AGA Community Roundtable to receive updates.