1. With one-time application, FIT tests are approximately 80% sensitive for cancer detection and approximately 20%-30% sensitive for advanced neoplasia detection. To enhance advanced adenoma detection, repeated applications of FIT are required. Therefore, the task force recommends repeated testing (see later for details) to maximize the effectiveness of cancer detection and prevention with this modality. Individuals choosing FIT should understand the need for recurring testing and for colonoscopy to evaluate a positive FIT result. Programs to track cycles of testing are encouraged to facilitate completion.
2. Given the high positive predictive value of FIT for cancer detection, colonoscopy is recommended when the test is positive, not repeat FIT.
3. When comparing FIT with guaiac-based fecal occult blood test, FIT has improved sensitivity for CRC and advanced colorectal neoplasia detection at similar levels of specificity. There is RCT-level evidence that adherence is superior for single-sample FIT compared with traditional 3-card gFOBT. Given these advantages, the task force recommends the use of FIT over gFOBT.
4. Based on currently available evidence, including the systematic reviews discussed earlier, the task force suggests a 1-sample annual FIT screening approach.
5. Performance characteristics of quantitative and qualitative FITs for neoplasia appear generally similar. However, the task force suggests that quantitative FITs be selected over qualitative FITs. This recommendation is based on improved quality control with automated reading and the ability to adjust fecal hemoglobin cut-off concentrations to define a positive test.
6. The optimal cut-off value for FIT should be determined by its performance characteristics, cost effectiveness, FIT device, and the screening program’s available colonoscopy resources. Based on the limited evidence, the task force favors a lower threshold cut-off FIT (ie, ≤20 μg/g) to define a positive test. The decision to recommend use of FIT with a hemoglobin threshold including 20 μg/g (not just less than that threshold) reflects, in part, a practical consideration because that threshold currently is used by the commonly available quantitative test in the U.S.
7. When FIT is positive in screen-eligible individuals, colonoscopy is the recommended test for subsequent evaluation.
8. The task force suggests that in the absence of iron-deficiency anemia or signs or symptoms of upper gastrointestinal pathology, a positive FIT and a negative colonoscopy should not prompt upper gastrointestinal evaluation.
9. There is no rationale to adjust diet or anticoagulation or antiplatelet agents when using FIT-based screening. The task force recommends that, to simplify testing and enhance adherence, patients should be instructed explicitly that they do not need to adjust diet or medications to complete a FIT test.
10. There is limited information examining the test characteristics of FIT when applied to a stool specimen obtained by digital rectal examination. Available data suggest that test characteristics may suffer. The task force suggests that FIT screening programs rely on spontaneously passed stool specimens and not an in-office digital rectal examination sample.
11. Although limited data have indicated that ambient temperature affects test positivity, current evidence is insufficient to recommend against distributing or mailing FITs when outside temperatures are above a certain level. Programs using FIT should adhere closely to test manufacturer’s specifications regarding storage and transport to minimize the effect of sample instability on FIT performance.
12. There is no strong evidence that delays in FIT kit return of up to 10 days after sample deposit affects FIT performance. Nonetheless, the task force suggests that participants in FIT-based programs should be informed about the importance of rapid return of the kit (ie, preferably mailing it or returning it to the laboratory within 24 hours) once the sample has been deposited. Furthermore, programs should establish quality-assurance practices to monitor return times of the FIT kits and solicit repeat samples when kits fall outside the predetermined range of acceptability based on the device used (as established by the manufacturer).
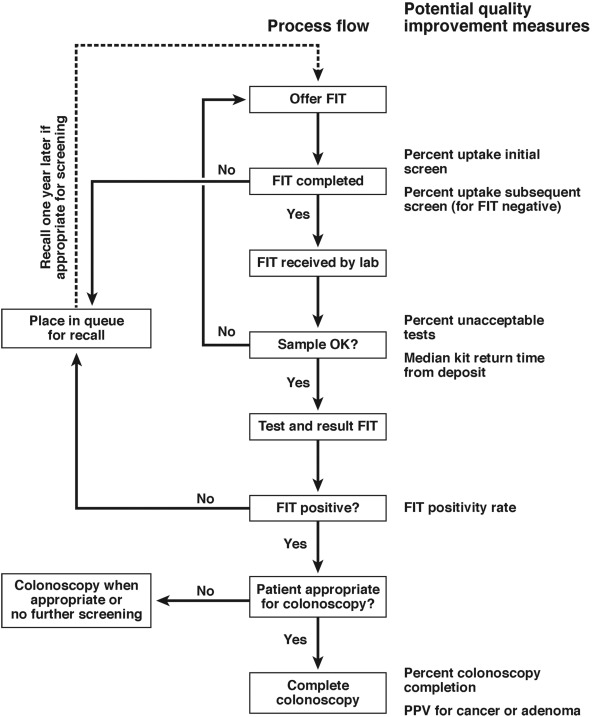
13. Similar to colonoscopy-based programs, FIT-based screening programs require careful attendance to quality assurance in provision of the test. Studies showing improved outcomes for selected measures in this area are needed. As this information is being developed, the committee suggests the following quality metrics for FIT-based testing programs:
a. FIT completion rate to those offered testing of 60% or greater.
b. Proportion returning FIT that cannot be processed by the laboratory of less than 5%.
c. Colonoscopy completion rate for those with a positive FIT of 80% or greater.
d. Adenoma detection rate greater than 45% in men and 35% in women on colonoscopy examinations performed to evaluate a FIT-positive test that uses a hemoglobin threshold of 20 μg/g or less.