Bakul Patel, MSEE, MBA
Director
Digital Health Center of Excellence
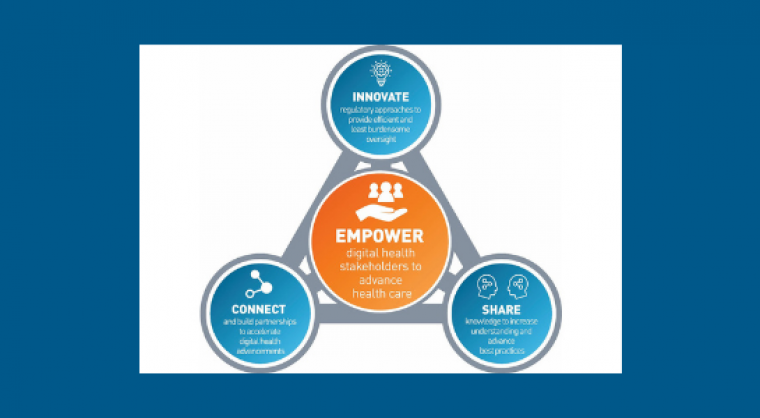
In September 2020, the Food and Drug Administration (FDA) announced the launch of the Digital Health Center of Excellence (DHCoE) within the Center for Devices and Radiological Health. Bakul Patel, MSEE, MBA, has been appointed as the DHCoE’s first director. Mr. Patel leads the DHCoE in advancing health care by promoting high-quality health innovations. This goal of the center under Mr. Patel’s leadership aims to empower stakeholders by connecting and building partnerships, share knowledge and innovate the regulatory approached needed to meet the prescribed FDA standards for safe and effective products.
Mr. Patel will give the closing keynote lecture at the 2021 AGA Virtual Tech Summit, the signature meeting of the AGA Center for GI Innovation and Technology, on April 30. In the following Q&A, Mr. Patel shares his perspectives on the goals of the DHCoE and its future direction.
Why was the DHCoE established?
The Digital Health Center of Excellence was launched as a part of the planned evolution of the Digital Health Program in the Center for Devices and Radiological Health. The announcement of the launch of the DHCoE marks the beginning of a comprehensive approach to digital health technology, setting the stage to advancing and realizing the potential of digital health.
What role do you see GI medical societies playing in the center’s Strategic Partnerships?
The Digital Health Center of Excellence aims to connect and build partnerships to accelerate digital health advancements; share knowledge to increase awareness and understanding, drive synergy, and advance best practices; and innovate regulatory approaches to provide efficient and least burdensome oversight. GI medical societies can play a role in providing the clinical feedback and health care provider perspective and insights on using different types of digital health technologies such as wearables and AI/ML (artificial intelligence/machine learning) devices.
Where do you see the DHCoE in a post-COVID era?
The pandemic has accelerated the arrival of the digital era in many aspects of American health care. The FDA will continue to learn and iterate to advance regulatory science for digital health advancement in accordance with emerging evidence and stakeholder feedback, remaining committed to its goal of supporting patient-centered innovations that are safe and effective.
Learn more about the DHCoE
- Kadakia, K., Patel, B., Shah, A. Advancing digital health: FDA innovation during COVID-19. NPJ Digit Med. 3(1):161 (2020)
- https://www.fda.gov/medical-devices/digital-health-center-excellence
About the 2021 AGA Virtual Tech Summit
The AGA Tech Summit, organized by the AGA Center for GI Innovation and Technology, brings together innovators, entrepreneurs, clinicians, medical technology and device companies, venture capitalists and regulatory agencies, to discuss the development and adoption of new innovations in gastroenterology. The eleventh annual meeting will take place virtually on April 29 and 30, 2021. Learn more at www.gastro.org/agatech.