Home > Advocacy & Policy > Policies Affecting GI > Regulatory relief: Step therapy threatens patients’ access to treatment
Regulatory relief: Step therapy threatens patients’ access to treatment
Step therapy, also known as “fail first,” is utilized by insurers to determine drug coverage and requires patients to try and fail on insurers’ preferred medications before covering the initial therapy prescribed by their health care provider.
AGA position: Provide a transparent appeals process for patients subjected to step therapy protocols to ensure timely access to care.
Step therapy requires patients to take medications they may have already tried and failed or have had adverse effects from in order to step through to the physician recommended treatment.
Step therapy exposes GI patients to less safe medications.
Based on a 2015 study, 18 major health plans representing 97 million lives required 45 percent of patients who rely on biologics or immunologic drugs to step through one or more therapies with black box safety warnings before they were able to access a safer treatment.1
With the increase of biologics to treat diseases like inflammatory bowel disease (IBD), more and more patients with digestive diseases are subject to this policy and potential delays to medically-appropriate care.
Step therapy is on the rise among private payors with questionable patient outcomes.
In 2018, CMS issued a policy change and began to allow Medicare Advantage (MA) plans to use step therapy protocols on Part B drugs. With one in three Medicare beneficiaries on an MA plan, this policy has the potential to greatly impact patient outcomes and health care costs in the Medicare program.
This legislation provides a clear and timely appeals process when a patient is subjected to step therapy. Specifically this legislation:
Establishes a clear exemption process
Requires insurers to implement a clear and transparent process for physicians or patients to request an exception to a step therapy protocol.
Outlines five exceptions to fail first protocols
Requires insurers to grant an exception if an appeal clearly demonstrates any of the following:
- Patient already tried and failed on the required drug.
- Delayed treatment will cause irreversible consequences.
- Required treatment is contraindicated and will/is likely to cause an adverse reaction.
- Required treatment will/is expected to prevent a patient from working or fulfilling activities of daily living.
- Patient is stable on their current medication.
Requires an insurer to respond to an exemption request within 72 hours under normal circumstances, and within 24 hours if life threatening
More problems with step therapy
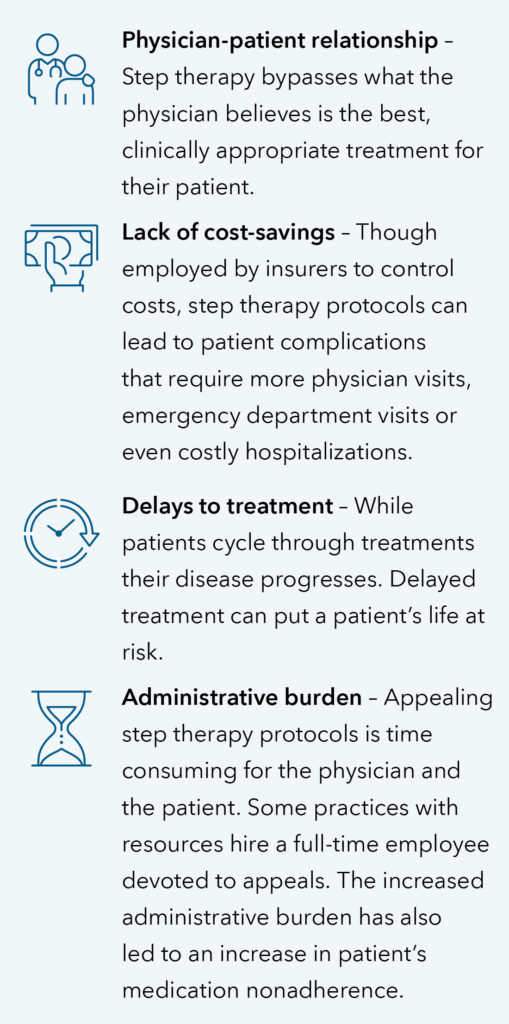
Bottom line: The Safe Step Act does not eliminate step therapy but provides exceptions when warranted. AGA urges Congress to enact the legislation so that patients can access the most effective treatment in a timely manner and physicians are able to decide the best treatment for their patients.
1. Branning, G., et al. “Formulary Management of Branded Drugs With And Without Boxed Warnings Within Therapeutic Categories.” Value in Health 18.3 (2015): A100.











